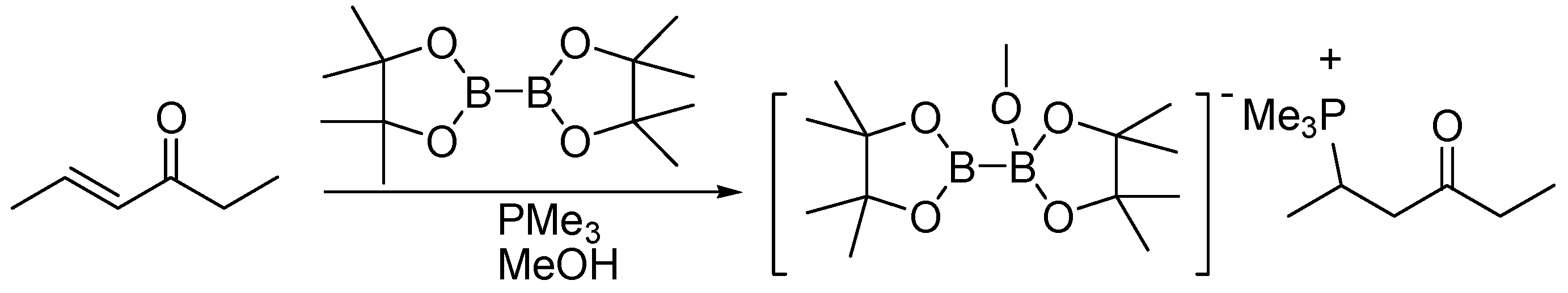
Palladium-Mediated Dealkylation of N -Propargyl-Floxuridine as a Bioorthogonal Oxygen-Independent Prodrug Strategy | Scientific Reports

Recent strategies in the synthesis of thiophene derivatives: highlights from the 2012–2020 literature | SpringerLink
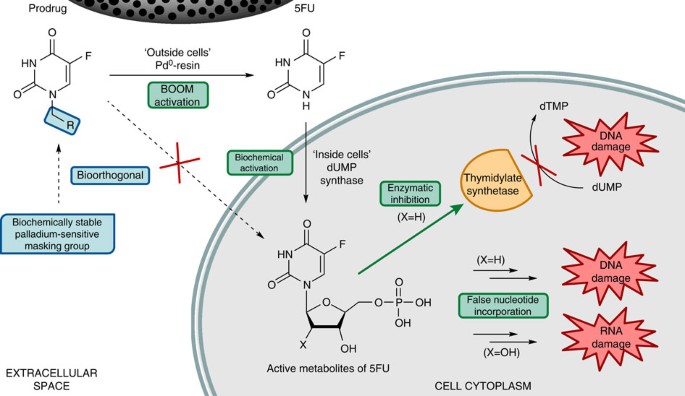
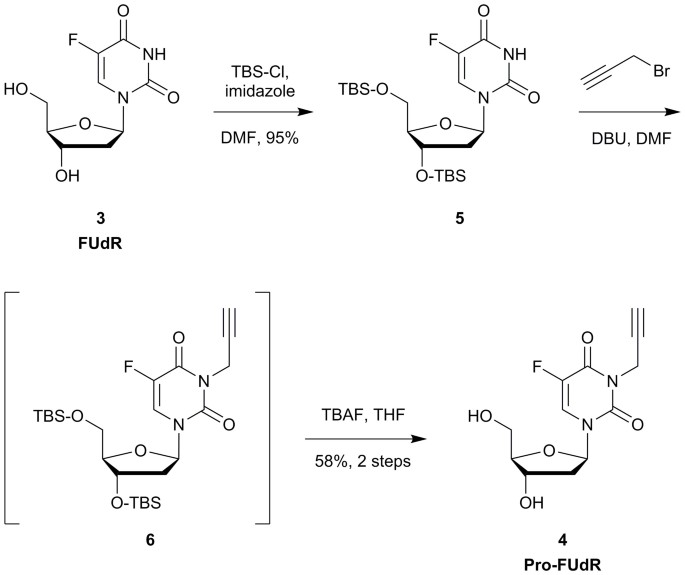
Extracellular palladium-catalysed dealkylation of 5-fluoro-1-propargyl-uracil as a bioorthogonally activated prodrug approach | Nature Communications
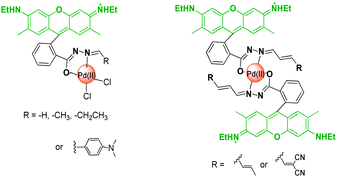
Colourimetric and fluorescent probes for the optical detection of palladium ions - Chemical Society Reviews (RSC Publishing)
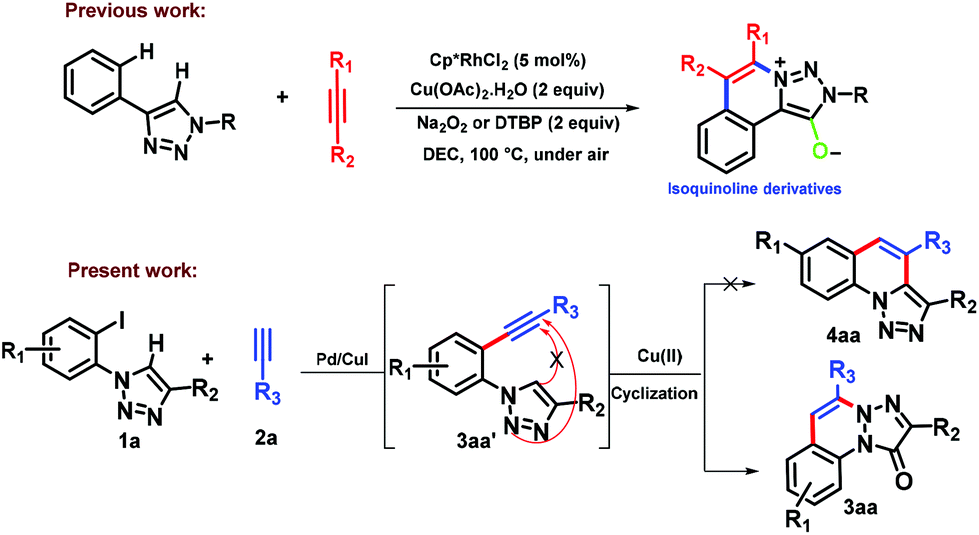
Unprecedented synthesis of 1,2,3-triazolo-cinnolinone via Sonogashira coupling and intramolecular cyclization - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB01152D
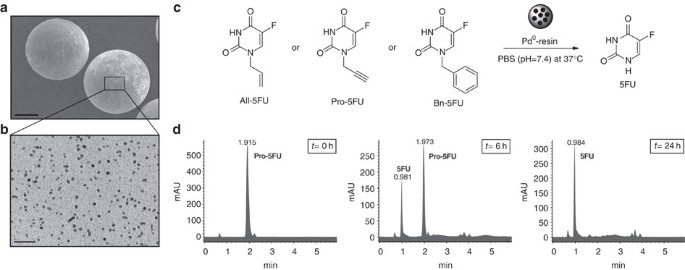
Extracellular palladium-catalysed dealkylation of 5-fluoro-1-propargyl-uracil as a bioorthogonally activated prodrug approach | Nature Communications
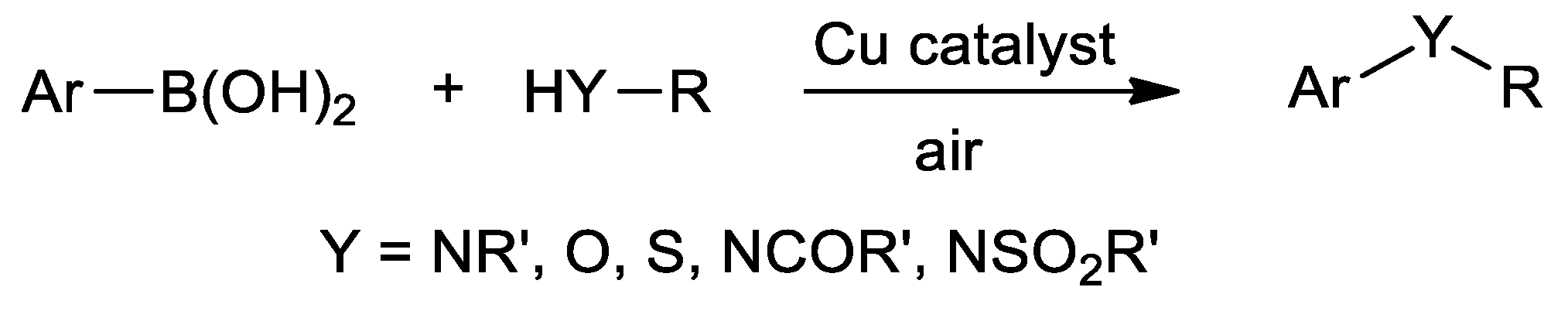
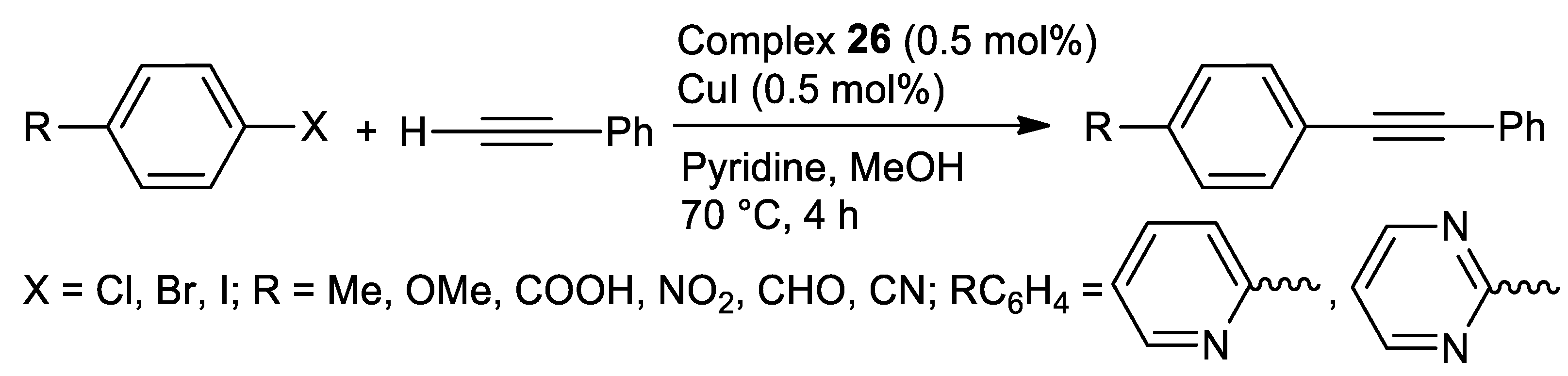
Catalysts | Free Full-Text | Thiosemicarbazone Complexes of Transition Metals as Catalysts for Cross-Coupling Reactions | HTML
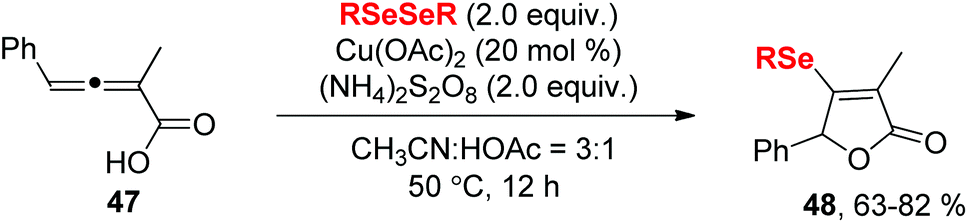
Recent advances in tandem selenocyclization and tellurocyclization with alkenes and alkynes - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D0QO00849D

Frontiers | Raising the Diversity of Ugi Reactions Through Selective Alkylations and Allylations of Ugi Adducts | Chemistry

Diazo Compounds: Versatile Synthons for the Synthesis of Nitrogen Heterocycles via Transition Metal‐Catalyzed Cascade C–H Activation/Carbene Insertion/Annulation Reactions - Xiang - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library

Catalysts | Free Full-Text | Thiosemicarbazone Complexes of Transition Metals as Catalysts for Cross-Coupling Reactions | HTML
Catalysts | Free Full-Text | Thiosemicarbazone Complexes of Transition Metals as Catalysts for Cross-Coupling Reactions | HTML