Pyrazol-1-yl)carbonyl palladium complexes as catalysts for ethylene polymerization reaction - ScienceDirect
Palladium(II) carboxylates and palladium(I) carbonyl carboxylate complexes as catalysts for olefin cyclopropanation with ethyl diazoacetate - Dalton Transactions (RSC Publishing)

Asymmetric carbonyl-ene reaction of trifluoropyruvate catalyzed by Pd(II)-SunPhos complex - ScienceDirect

Direct Dehydrogenation for the Synthesis of α,β‐Unsaturated Carbonyl Compounds - Chen - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library
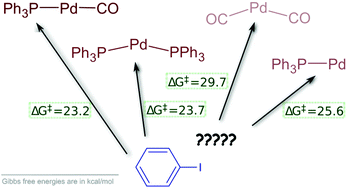
Viable pathways for the oxidative addition of iodobenzene to palladium (0)-triphenylphosphine-carbonyl complexes: a theoretical study - Dalton Transactions (RSC Publishing)

Synthesis, characterization and CO-releasing property of palladium(II) bipyridine flavonolate complexes | SpringerLink

Proposed mechanism for the palladium pincer-catalyzed carbonylative... | Download Scientific Diagram

PDF) Palladium(I) and palladium(0) carbonyl bromide complexes | Vitaly Belsky and Oleg Temkin - Academia.edu

Stereoselective Carbonyl Allylation by Umpolung of Allylpalladium(II) Complexes - Zanoni - 2007 - European Journal of Organic Chemistry - Wiley Online Library
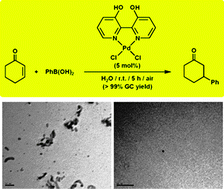
Conjugate addition of arylboronic acids to α,β-unsaturated carbonyl compounds in aqueous medium using Pd(ii) complexes with dihydroxy-2,2′-bipyridine ligands: homogeneous or heterogeneous nano-catalysis? - Catalysis Science & Technology (RSC Publishing)

Binuclear Palladium Complex Immobilized on Mesoporous SBA-16: Efficient Heterogeneous Catalyst for the Carbonylative Suzuki Coupling Reaction of Aryl Iodides and Arylboronic Acids Using Cr(CO) 6 as Carbonyl Source | SpringerLink
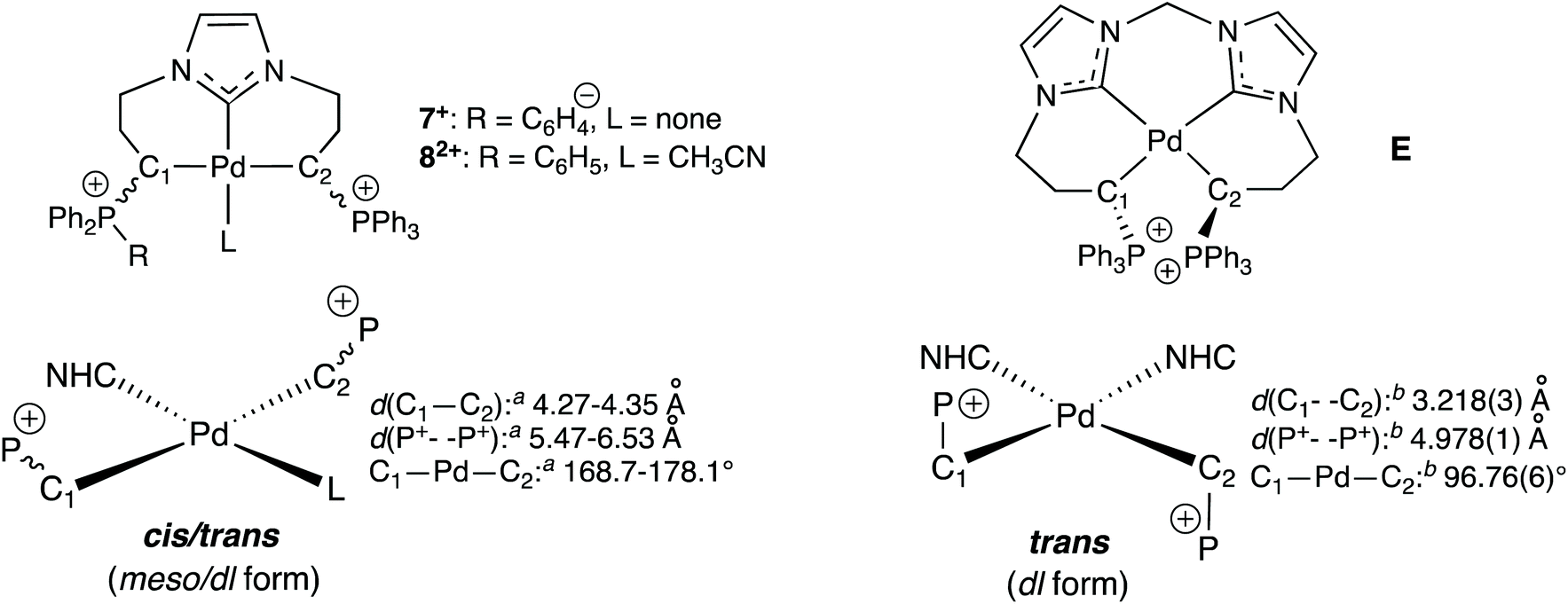
Palladium(ii) pincer complexes of a C,C,C-NHC, diphosphonium bis(ylide) ligand - Dalton Transactions (RSC Publishing)

SYNTHESIS, CHARACTERIZATION, AND CYTOTOXICITY OF MIXED-LIGAND COMPLEXES OF PALLADIUM(II) WITH 1,10-PHENANTHROLINE AND N-CARBONYL-L-ISOLEUCINE DIANION - тема научной статьи по химии из журнала "Координационная химия"
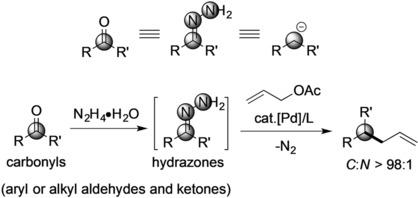
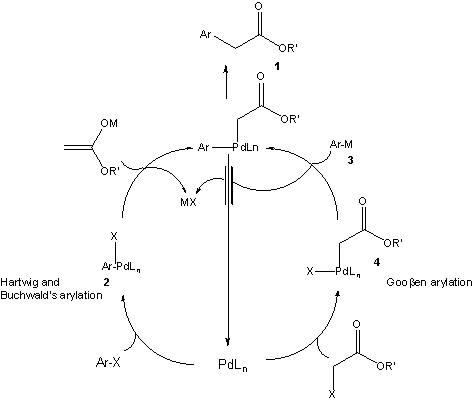
Umpolung of Carbonyl Groups as Alkyl Organometallic Reagent Surrogates for Palladium‐Catalyzed Allylic Alkylation - Angew. Chem. Int. Ed. - X-MOL


![IR carbonyl stretching frequencies of [Rh(CO) 2 Cl(L)] complexes... | Download Table IR carbonyl stretching frequencies of [Rh(CO) 2 Cl(L)] complexes... | Download Table](https://www.researchgate.net/publication/309115781/figure/tbl1/AS:667603408285696@1536180464134/IR-carbonyl-stretching-frequencies-of-RhCO-2-ClL-complexes-bearing-various.png)